On Monday, we kicked off our series on using Wolfram|Alpha for chemistry in honor of the American Chemical Society’s Fall 2009 National Meeting & Exposition, taking place in Washington, DC, USA this week. In this post, we begin to break down chemistry topics by taking a look Wolfram|Alpha’s collection of chemical element data. If you are attending the meeting, stop by the Wolfram Research booth, #2101, for a personal introduction to Wolfram|Alpha and the technology behind it.
The periodic table and its elements can be viewed as the foundation for building your knowledge and understanding of chemistry. Wolfram|Alpha defines a chemical element as any of the more than 100 known substances that cannot be separated into simpler substances and that singly or in combination constitute all matter. Currently, there are 118 commonly recognized elements, 92 of which occur naturally, and the others synthetically. The periodic table is organized in 18 columns (called groups) and 7 rows (called periods). Elements are arranged in the table based on their atomic weight.
In Wolfram|Alpha you can retrieve data for a chemical element in a number of ways, such as by name, symbol, atomic number, or a specific class, such as radioactive elements. In this example we query “hydrogen” and quickly learn from the basic elemental properties pod that it has an atomic number of one, which places it in the first position on the periodic table. We also learn its symbol, atomic weight, thermodynamic properties, material properties, electromagnetic properties, reactivity, atomic properties, abundances, nuclear properties, and identifiers. Click the image to explore more properties of hydrogen.
In addition to calling up an element, you can request specific information such as the number of protons, neutrons, and electrons in an element; compare properties of two elements; compare properties for classes of elements; do computations using properties of chemical elements; or call up elements that meet a specific criteria. In this example, we ask Wolfram|Alpha to compute the ten densest elements:
Wolfram|Alpha also lets you explore the elemental abundance of the universe by ranking the most common elements:
Are you a professional who is using Wolfram|Alpha in your research today? Are you an instructor who has incorporated Wolfram|Alpha in the classroom, or are you a student looking for more ways to incorporate Wolfram|Alpha into your chemistry studies? Share your experiences with other chemistry enthusiasts having this conversation on the Wolfram|Alpha Community site.
Cool, but the universal abundance of elements croaks on simple things like earth, venus and other planets.
What do you mean? Are you talking about elemental abundance on different planets? That’s quite different from universal abundance…
Funny and interesting to you system.
Learned about it by accident from a review of one of the magazines for webmasters. I was immediately interested in the way of its potential and opportunities .. – Maybe two hours I spent for computing the right formula to me – but your system did it all for a couple of minutes. Thank you – that have created this service, I hope, over time, it will become a worthy competitor to all the well-known search engine Google.
I wish you good luck!
PS
It is easier to forgive, I do not know English, so had to use an interpreter. I hope I was able to convey to you the meaning of my words.



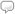 Comments Off
Comments Off


